Project management services
- UCLB Project Managment
- Regulatory services
- Prototyping services
- Product design services
- Commercial and technical risk assessment services
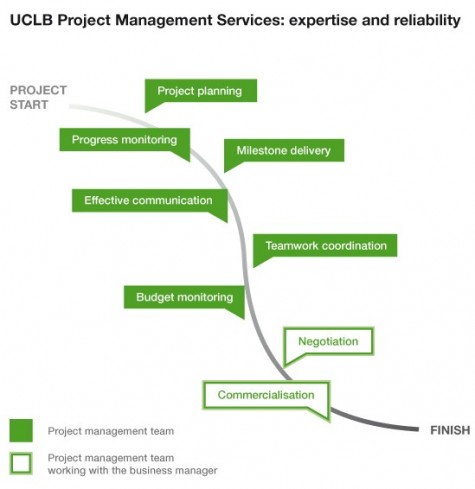
At UCLB we can provide departments and institutes with a comprehensive and friendly project management service.
Our dedicated project managers are fully qualified and equipped with the appropriate knowledge, contacts, expertise and experience to support either substantive single or multiple stakeholder projects.
For a negotiated fee our project managers work closely with UCLB Business Managers to proactively assist the principal investigators and clinicians in managing the full life cycle of the project, as well as contributing to teamwork within the group. They will also maintain effective liaison with key people and organisations, both internal and external, to ensure that projects are delivered according to plan, on time and within budget.
For all your project management needs UCLB Project Management Services are happy to offer an expert, reliable and professional service. Find a list of our Project Management team in Our People page.
The Project Management team has an excellent track record of successfully CE Marking new novel medical devices of all classes under its ISO accredited Quality Management System.
This involves:
- Technical file compilation and submission to Notified Body
- Obtain CE mark or 510k approval for a device
- Support the maintenance of the technical file
- Provide the client with the necessary information and support to place the product on the market
- Clinical Effectiveness Trials
Once the design has been finalised the Project Management team works closely with all stakeholders to ensure that the prototype is delivered as efficiently as possible.
These activities include:
- Product specification
- Production of prototypes
- Testing of prototypes, both software and hardware devices
The Project Management team works with a large number of designers, manufacturers and test facilities in all areas of medical device development to ensure that we can deliver a high quality, efficient service to our clients. The team coordinates all activities to ensure that all regulatory requirements have been incorporated into the device development.
Activities include:
- Regulatory assessment
- Product specification
- Design drawings
- CAD files
- Material specifications of the device
The Project Management team has the knowledge and expertise to compile full technical and commercial risk assessments for our clients.
These assessments include:
- Commercial and technical risk assessment of the concept
- Evaluation of intellectual property status
- Initial specification of user requirements
- Market analysis
- Regulatory pathway mapping both for CE marking and 510K approval